Abstract
Background:
Coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is characterised by a hyperinflammatory and prothrombotic state. In the most severe cases, patients can develop lung complications such as pneumonitis and acute respiratory distress syndrome, frequently complicated by venous and arterial thrombosis. Recent studies suggest that platelets play a role as immune mediators, and may therefore contribute to the development of hyperinflammation and thrombosis in severe COVID-19 patients.
The expression and signalling of Glycoprotein VI on platelets are dependent on its association and cross-linking with the dimeric Fc receptor γ-chain, which, upon platelet activation, leads to a signalling cascade involving the Spleen Tyrosine Kinase (SYK). Fostamatinib, a potent SYK inhibitor, has been approved for treatment of Immune Thrombocytopenia (ITP), and has been implicated in abrogating thrombosis by mitigating SYK signalling in platelets. We have recently shown that SYK inhibition can abrogate the thrombosis caused by cross-linking of COVID-19 antibodies.
In this study, we assessed platelet reactivity using the Platelet Phenomic Analysis (PPAnalysis), which identifies platelet sensitivity and functional response as independent measures of platelet reactivity by flow cytometry. We hypothesised that platelets in patients with severe COVID-19 are hypersensitive to platelet agonists, in particular to the collagen-related peptide agonist, which involves the SYK pathway of platelet activation.
Methods:
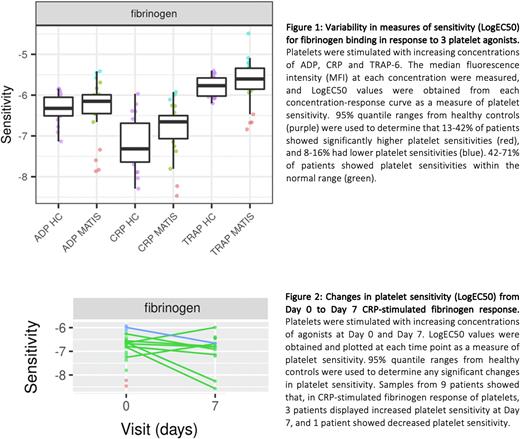
Patients with COVID-19 pneumonia, recruited to the Multi-Arm Trial of Inflammatory Signal inhibitors for COVID-19 (MATIS) clinical trial - an interventional study assessing the impact of Fostamatinib (FOS) or Ruxolitinib (RUX) against Standard of Care (SOC) to prevent escalation of COVID-19 from moderate (WHO grades 3 and 4) to severe (WHO grade 5) - were included in this study, REC reference 20/HRA/2618. Samples were collected in citrated vacutainers, taken at day 0, before starting study medication or SOC, and at days 7, 14 and 28, where possible. Samples were processed immediately to obtain platelet-rich plasma (PRP). PRP was labelled with fluorescein isothiocyanate (FITC)-conjugated anti-fibrinogen and PE-Cy5-conjugated P-Selectin antibodies, and stimulated with increasing concentrations of the platelet agonists, adenosine diphosphate (ADP; 0.03-30 µM), collagen-related peptide (CRP; 0.003-3 µg/mL), and thrombin receptor activator peptide 6 (TRAP-6; 0.05-15 µM). Changes in platelet sensitivity, measured as LogEC50, were analysed using R.
Results:
40 of 182 patients recruited into MATIS were included. A total of 50 COVID-19 samples (24 at day 0, 14 at day 7, 2 at day 14, and 10 at day 28) and 59 healthy control samples were processed using PPAnalysis. The 24 samples taken at day 0 showed extensive variability in platelet sensitivities between the responses to 3 agonists, with outliers both above and below the 95% centile of controls: 0-4% (P-Selectin response) and 13-42% (fibrinogen response) of patients displayed higher platelet sensitivity (below the 95% centile for controls), and 4-42% (P-Selectin response) and 8-16% (fibrinogen response) had lower sensitivity (above the 95% centile for controls), (Figure 1). Of the 9 patients with samples also taken at day 7, no patients showed an increase in platelet sensitivity from the P-Selectin response to all agonists. 5 patients showed reduced sensitivity. 2 patients in this cohort required non-invasive ventilation (NIV). Conversely, fibrinogen response to all agonists showed increased sensitivity at day 7 in 6 patients, with 1 patient requiring NIV, and reduced sensitivity was observed in 2 patients, both requiring NIV (Figure 2).
Conclusions:
Patients with severe COVID-19 display a large variability in platelet function, with either higher or lower platelet sensitivity to the agonists at day 0 when compared to controls. This implicates a role for aberrant platelet reactivity in COVID-19 and its associated prothrombotic state, and could contribute to the morbidity and mortality of patients. Interpretation of day 7 results at present is limited due to small numbers. However, it does show utility of the PPAnalysis, as a useful tool in the stratification of different patient cohorts, and in studying the associations between platelet function and disease.
Disclosures
Cooper:Sanofi, Principia, Novartis, Griffols, Sobi, Argenyx, UCB, Rigel: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal